Phosphoric Acid and Lithium Iron Phosphate Battery
Phosphoric acid is a key raw material in the production of lithium iron phosphate (LFP) batteries, which are widely used as cathode materials in energy storage systems and electric vehicles.
In the phosphate group, a phosphorus atom is coordinated with four oxygen atoms to form a tetrahedral PO₄ structure, with phosphorus at the center. The crystal framework of lithium iron phosphate consists of FeO₆ octahedra and PO₄ tetrahedra arranged alternately to form a distinctive Z-shaped chain structure. Lithium ions occupy octahedral sites within this shared-edge framework. In the lattice, FeO₆ octahedra are connected through shared corners in the bc plane, while LiO₆ octahedra form edge-sharing chains along the b-axis. Each FeO₆ octahedron shares edges with two LiO₆ octahedra and one PO₄ tetrahedron, whereas each PO₄ tetrahedron shares edges with one FeO₆ octahedron and two LiO₆ octahedra.
From a crystallographic perspective, lithium iron phosphate belongs to the orthorhombic crystal system with the space group Pmnb. The lattice parameters are a = 6.008 Å, b = 10.334 Å, and c = 4.693 Å, with a unit cell volume of 291.4 ų. The presence of stable phosphate groups reinforces the crystal framework, giving the material excellent thermal stability and long cycle life.
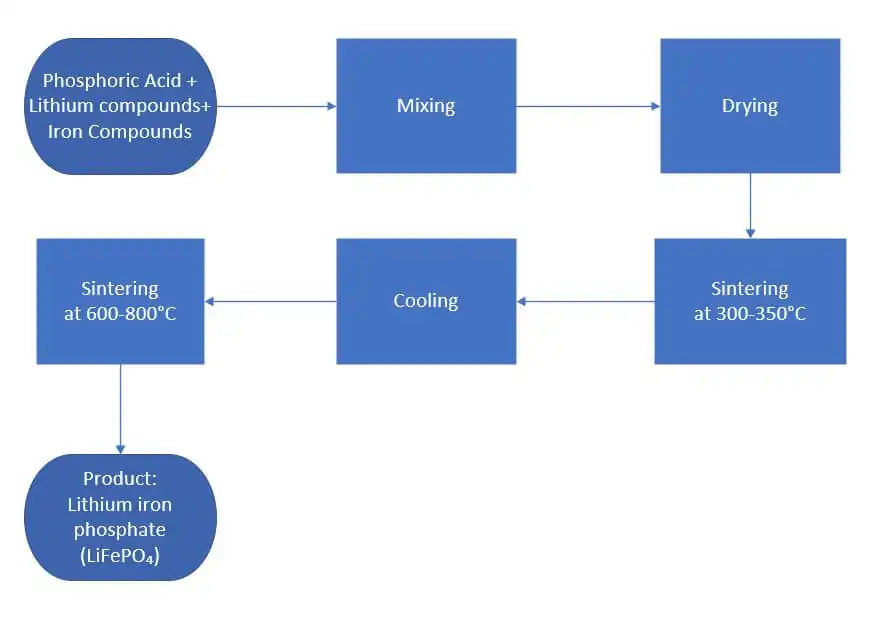
Among various synthesis routes, the high-temperature solid-state reaction method is the most mature and widely adopted technique for producing lithium iron phosphate. In this process, iron, lithium, and phosphorus sources are uniformly mixed according to the stoichiometric ratio and then dried. The mixture is first calcined at a relatively low temperature of 300–350 °C for 5–10 hours under an inert atmosphere to decompose the precursors.
This is followed by high-temperature sintering at 600–800 °C for 10–20 hours to obtain olivine-structured lithium iron phosphate.
Lithium Iron Phosphate (LFP) Cathode Material
Phosphoric acid is a critical raw material in the production of lithium iron phosphate (LFP), a widely used cathode material for lithium-ion batteries in electric vehicles and energy storage systems.
LFP features a stable olivine crystal structure composed of iron-oxygen (FeO₆) octahedra and phosphate (PO₄) tetrahedra. In this structure, each phosphorus atom is bonded to four oxygen atoms, forming a robust PO₄ tetrahedron. These FeO₆ octahedra and PO₄ tetrahedra are alternately connected, creating a rigid three-dimensional framework, while lithium ions occupy octahedral sites within the lattice. This highly ordered structure effectively stabilizes lithium ion movement during charge and discharge cycles.
Crystallographically, lithium iron phosphate belongs to the orthorhombic crystal system with a Pmnb space group. The lattice parameters are a = 6.008 Å, b = 10.334 Å, and c = 4.693 Å, with a unit cell volume of 291.4 ų. The strong phosphate framework significantly enhances structural stability, resulting in excellent thermal safety, long cycle life, and reliable electrochemical performance.
Manufacturing Process
Lithium iron phosphate is primarily produced using a high-temperature solid-state reaction process, which is recognized as the most mature and scalable manufacturing method. Precisely measured iron, lithium, and phosphorus raw materials are uniformly mixed according to stoichiometric ratios and thoroughly dried. The mixture undergoes a low-temperature pre-calcination at 300–350 °C under an inert atmosphere to decompose the precursors, followed by high-temperature sintering at 600–800 °C to form high-purity, olivine-structured lithium iron phosphate.
Key Advantages
-
Excellent thermal and chemical stability
-
Long cycle life and consistent performance
-
High safety for electric vehicle and energy storage applications
-
Mature, reliable production process